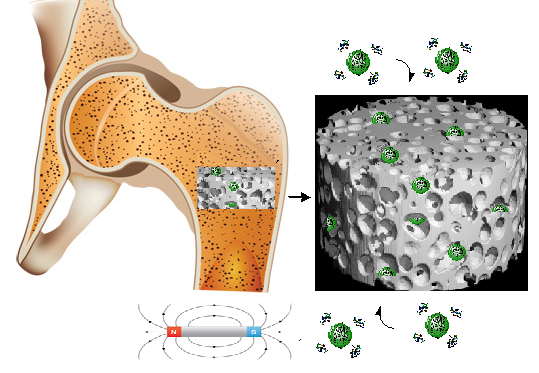
Current clinical treatments used for large bone defects are challenging despite the natural capacity of bone for healing. For critical injuries (critical size defect) it is required invasive surgical intervention to aid bone regeneration. Creating of 3D scaffold with controlled internal architecture have to be correlated with multiple stimuli, contributors or mediators of bone regeneration and de novo tissue formation, including physical and biochemical factors. The next generation of scaffolds and tissue engineered constructs need to be driven by a personal medicine approach and must integrate a range of biological and physical properties for optimal bone regeneration. In this context the main concept of the project is to optimize and scale-up the magnetic scaffolds obtained via biomimetic methods as alternative for actual regenerative medicine for bone repair. The new therapy involves the use of magnetic scaffolds as a stimuli-responsive matrix (biological signals, presence of growth factors, drugs or other bio-agents). The complex 3D system proposed in project has potential clinical application as bone graft materials to fill the void obtained after the surgicals. Incorporating superparamagnetic nanoparticles (MNPs) into 3D scaffolds they are sensitive to magnetic field and can be loaded with biological agents and release them as a response to physical stimulus. The objectives of the project are: to evaluate the 3D biomimetic scaffolds able to respond to biological and physical stimuli as osteoiductive and osteoconductive matrix (scientific impact); improve the technology for a magnetic osteoinductive scaffold (technological impact); therapy based on tests which simulate the applicative condition (economical impact, transfer from TRL3 to TRL4); Increasing the qualification of human resources by stimulation of professional training on the new advanced therapies (social impact).